Malaria vaccines provide strong and lasting immunity
by NIH
Antimalarial drugs and measures to control mosquitoes have reduced the malaria burden worldwide. But this decline has stalled in recent years. Further progress against malaria will likely require vaccines, which have so far conferred only partial protection.
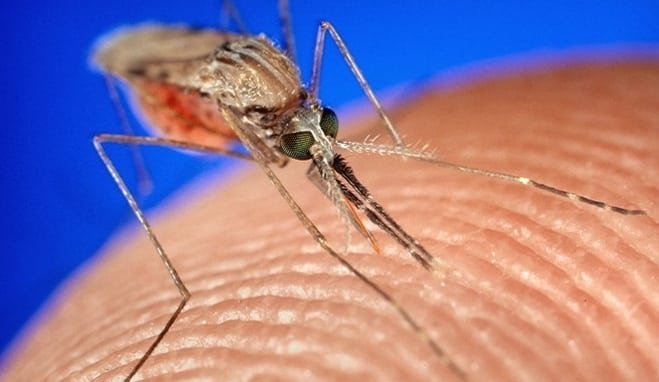
Malaria is caused by single-celled parasites from the genus Plasmodium. Infected mosquitoes transmit these parasites to people in a form called a sporozoite. Upon infection, sporozoites travel to the liver. There, they grow and replicate before they enter the blood and infect red blood cells. Only when the parasites infect red blood cells do they cause disease.
One potential vaccination strategy combines live sporozoites with an antimalarial drug. The sporozoites induce an immune response while the drugs kill the sporozoites before they can cause disease. This is called chemoprophylaxis vaccination, or CVac. Previous trials found that CVac conferred high immunity to malaria. But this immunity extended only to the specific parasite variant used in the vaccine.
A research team led by Dr. Patrick Duffy of NIH’s National Institute of Allergy and Infectious Diseases (NIAID) and Dr. Stephen Hoffman of Sanaria Inc. further explored the CVac approach in Phase 1 clinical trials. Results were published in Nature on June 30, 2021.
Healthy volunteers were given three monthly doses of sporozoites and one of two drugs: pyrimethamine (PYR) or chloroquine (CQ). PYR kills liver-stage parasites before their release into blood, while CQ kills parasites at the blood stage. Three months after the last sporozoite dose, the researchers exposed the volunteers to malaria under carefully controlled conditions. In “homologous challenges,” volunteers were exposed to the same African variant as that used for vaccination. “Heterologous” challenges exposed volunteers to a distantly related variant from Brazil.

Carol graduated from Riverside White Cross School of Nursing in Columbus, Ohio and received her diploma as a registered nurse. She attended Bowling Green State University where she received a Bachelor of Arts Degree in History and Literature. She attended the University of Toledo, College of Nursing, and received a Master’s of Nursing Science Degree as an Educator.
She has traveled extensively, is a photographer, and writes on medical issues. Carol has three children RJ, Katherine, and Stephen – one daughter-in-law; Katie – two granddaughters; Isabella Marianna and Zoe Olivia – and one grandson, Alexander Paul. She also shares her life with her husband Gordon Duff, many cats, and two rescues.
ATTENTION READERS
We See The World From All Sides and Want YOU To Be Fully InformedIn fact, intentional disinformation is a disgraceful scourge in media today. So to assuage any possible errant incorrect information posted herein, we strongly encourage you to seek corroboration from other non-VT sources before forming an educated opinion.
About VT - Policies & Disclosures - Comment Policy



